Despite several studies many questions still remain regarding the water crisis in Flint, Michigan. A decision to not add a corrosion inhibitor to the city’s treated water appears to have been a key factor in the subsequent chain of events that resulted in a public health hazard.
Background: Switching the Public Water Source
On April 25, 2014, city officials in Flint, Michigan, changed the city’s water source from Lake Huron (purchased from and treated by Detroit, Michigan) to the Flint River, in an effort to save money. Flint’s existing water treatment plant was reactivated as part of the cutover.
As soon as the switch was made, brown discolored water began to come out of resident’s faucets. Within months, residents began reporting foul-smelling water and health issues such as rashes and hair loss. The city issued a boil-advisory to kill Escherichia coli bacteria lurking in the water. In response, the Flint water department added sodium hypochlorite (chlorine bleach) to kill the bacteria.
Unfortunately, the over chlorinated water reacted with organic matter to create toxic byproducts. Problems escalated six months later in October 2014, when a large automotive manufacturer in Flint expressed concerns that the high chlorine levels in Flint’s water would corrode its engine parts during the manufacturing process. (Learn how failures are preventable in the article Human Error as a Factor in Corrosion Failure.)
First Indications of Lead in the Water
Lead was discovered in the tap water after continued citizen complaints to city officials, and tests at some residences revealed a lead content of 104 ppb. In comparison, the US Environmental Protection Agency (EPA) allowable maximum is 15 ppb.
Lead is harmful because the body, tricked into treating lead like it would calcium, deposits lead in bones and in brain neurons. One medical study found that lead levels in children were higher after the water cutover.
How Lead Found Its Way into Flint’s Drinking Water
In almost all cases, lead is not present in the original water source or in the treatment facility. Instead, lead gets into drinking water from pipes and home water fixtures. The water acquires lead as it travels through the service pipes and home fixtures.
Water takes a long and convoluted route from the water treatment plant to the household faucet. After leaving the treatment plant, water is pumped through large water mains (made from iron) to neighborhoods. Smaller service lines carry the water to individual residences and commercial buildings.
Flint has approximately 20,000 lead service pipes. Many of the service lines, decades old, are made of lead because at one time there were laws and local ordinances in many cities that required the pipes to be made of lead.
Other sources of lead are found within the home, including lead in brass faucets and valves, galvanized iron, and the lead solder that is used to connect metal pipes together.
Protective Lead Scale Can Prevent Lead Poisoning
Many communities possess the same infrastructure as Flint, but do not have lead contaminated drinking water. This can be explained by understanding the chemical interactions that occur between the water and the pipes through which the water flows.
Shortly after lead pipes are installed, chemical reactions occur that cause a protective layer of lead scale to build up on the inside walls of the pipes (Figure 1). This protective barrier prevents lead from leaching into the water and prevents oxidants in the water from contacting the pipe’s surface. In essence, the scale, which consists of lead and other minerals, prevents the pipe's lead atoms from entering the water that is flowing in the pipe.
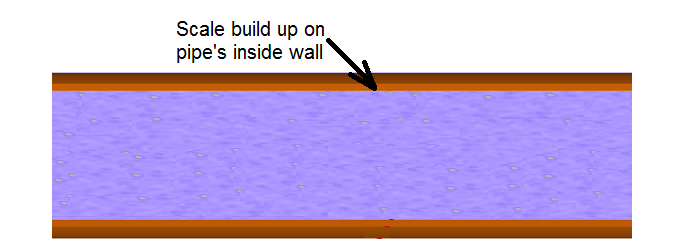
Figure 1. Inside section of pipe with scale build up.
To control pipeline corrosion, water treatment plants usually add a chemical corrosion inhibitor, such as phosphoric acid, polyphosphate or orthophosphate, which promote the buildup of scale to protect the water.
Some regulatory agencies (e.g., the EPA) have rules mandating the use of corrosion control in community water systems. At first it was thought that Flint’s water treatment plant used corrosion control chemicals, but officials later stated that it did not, presumably to reduce costs. Without corrosion inhibitors, tap water tends to contain much higher levels of lead and minerals. Experiments using water from Flint and Detroit showed that Flint’s water corroded an iron test sample faster than Detroit’s water.
Water from Flint’s treatment plant attacked the scale that normally prevents lead from leaching into the water. The deteriorating scale particles not only discolored the water, but also allowed the unprotected lead service pipes and lead fixtures to slowly dissolve into the water.
Legionnaires Disease
Complicating matters, lead contamination is not the only problem thought to be caused by a lack of corrosion inhibitors. An outbreak of Legionnaires Disease is thought to have infected 90 people and killed 12.
Normally, the chlorine that is added to the water as one of the final treatment steps kills the Legionnaires bacteria, among others. However, in Flint the breakpoint chlorination point was never reached, as the chlorine was thought to have reacted with the rusty iron-filled water and was used up, allowing the bacteria to flourish in the pipes.
Undoing the Damage to the Water System's Infrastructure
In October 2015, the city of Flint switched back to using water supplied by Detroit. It was estimated that it would take several months for protective scale to build up in the pipes. Numerous service lines and pipes are being replaced. The water is considered safe to drink after passing through a home tap filter. (Futher reading: Corrosion Costs & Recommended Practices for the Water Industry.) Thirteen criminal indictments were filed as a result of the crisis, and finger pointing and litigation is ongoing.