What Does
Polarization Resistance Mean?
Polarization resistance (Rp) is the transition resistance between the electrodes and the electrolyte. In other words, an increased resistance to the flow of current in a voltaic cell is caused by chemical reactions at the electrodes. Polarization results in a reduction of the electric potential across the voltaic cell.
An electrode is polarized when its potential is forced away from its value at open circuit or corrosion potential. Polarization of an electrode causes current to flow due to electrochemical reactions that it induces at the electrode surface.
Corrosionpedia Explains Polarization Resistance
Polarization is a deviation of the electrochemical process from equilibrium due to an electric current passing through the galvanic cell. Polarization may occur either at the cathode (cathodic polarization) or at the anode (anodic polarization). Polarization has three types, among them resistance polarization refers to the potential drop due to either the high resistivity of the electrolyte surrounding the electrode or an insulation effect of the film on the electrode surface formed by the reaction products.
Polarization resistance is defined by the following equation:

Where, 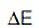 is a variation of the applied potential around the corrosion potential and
is a variation of the applied potential around the corrosion potential and 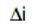 is the resulting polarization current. High Rp of a metal implies high corrosion resistance and low Rp implies low corrosion resistance. Thus, polarization resistance is the ratio of the applied potential and the resulting current response. This "resistance" is inversely related to the uniform corrosion rate.
is the resulting polarization current. High Rp of a metal implies high corrosion resistance and low Rp implies low corrosion resistance. Thus, polarization resistance is the ratio of the applied potential and the resulting current response. This "resistance" is inversely related to the uniform corrosion rate.
Testing polarization resistance is a useful technique that enables the estimation of a corrosion rate under steady-state conditions using Taefel slopes on the potentiodynamic scan. It is also used to describe the method of measuring corrosion rates using this slope.
Polarization resistance behaves like a resistor and can be calculated by taking the inverse of the slope of the current potential curve at open circuit or corrosion potential. During the polarization of an electrode, the magnitude of the current is controlled by reaction kinetics and diffusion of reactants both toward and away from the electrode.
During polarization measurement, several factors should be considered:
- Effects of scan rate
- Solution resistance
- Surface conditions
- Pitting potential
If these factors are not properly accounted for, they can cause misinterpretations and significantly affect the result.